Theseus Pharmaceuticals: Reinventing TKI’s – I’d Buy The Next Dip (NASDAQ:THRX)

Anastasia Yakovleva/iStock Editorial via Getty Images
Investment Overview
Back in January 2017, Japanese Pharma giant Takeda (TAK) agreed to buy the specialist oncology biotech Ariad Pharmaceuticals in a deal that valued Ariad at ~$5.2bn, or, $24 per share, which represented a ~75% premium to its traded price at the time.
Ariad’s lead drug candidate, the leukemia drug Iclusig, earned revenues of ~$260m for Takeda in fiscal year 2022, while a second Ariad candidate, brigatinib, an anaplastic lymphoma kinase (“ALK”) inhibitor indicated for lung cancer, is now approved under the brand name Alunbrig, earning ~$105m.
Today, four members of Ariad’s senior management team are working together at another biotech, Theseus Pharmaceuticals (NASDAQ:THRX) – former President Tim Clackson, former VP of Drug Discovery Bill Shakespeare, Former Senior Medical Director David Kerstein, and former VP of preclinical and translational research, Vic Rivera.
Theseus – the subject of this post – IPOd in October 2021, raising ~$160m at $16 per share. In a statement in the company’s Q322 10Q submission, the company describes itself as follows:
Our goal is to develop “pan-variant” kinase inhibitors—inhibitors that target all major cancer causing and drug resistance mutations in clinically significant protein kinases.
We believe that truly pan-variant inhibitors are required to effectively inhibit the heterogeneous mix of resistance mutations found in patients, and may also suppress the emergence of new mutations when used in earlier lines of therapy.
To develop such inhibitors, we deploy our novel predictive resistance assay, or PRA, a highly differentiated cell-based method for testing TKIs that we believe is predictive for “pan-ness.”
In a recent corporate presentation, the company discusses the three assets it currently has under development.

Theseus Pharmaceuticals pipeline (Corporate presentation)
Only lead asset THE-630 has made it into clinical trials to date, although the interim data from a Phase 1/2 dose escalation study ought to provide an intriguing catalyst for investors – or prospective investors – in Q223, as might the FDA’s response to the Investigational New Drug (“IND”) the company plans to submit for THE-349 in Q423.
Although Theseus shares leapt to a high of >$20 in the weeks following the company’s IPO, subsequent performance was disappointing – shares had fallen to a price >$5 by the end of 2022, representing a 75% decline – although not untypical of newly IPOd biotechs in 2022 – a savage bear market rocked almost biotech stocks in 2022.
My interest in Theseus is piqued for two main reasons, the first being the company’s innovative approach to the tyrosine kinase inhibitor class.
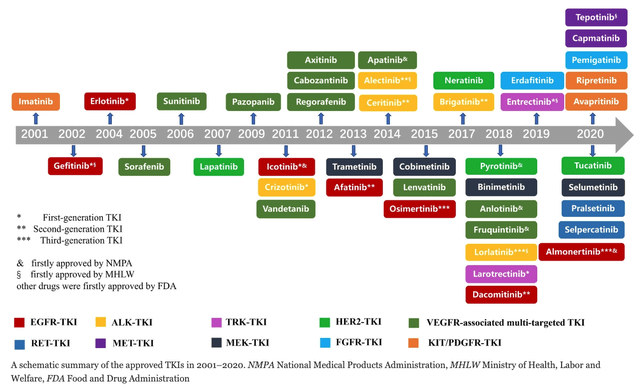
Approved TKI’s targeting solid tumors (biomedcentral)
As we can see in the diagram above from BioMedCentral a large number of TKIs have been approved since 2001 targeting a variety of solid tumors, and according to the University of Dundee:
It has been estimated that approximately 30% of current R&D spend in pharmaceutical companies is focused on the development of kinase inhibitors. To date 113 current approved drugs target kinases, comprising 103 kinase inhibitors (82 FDA approved) and 10 monoclonal antibodies (nine FDA approved). The current global market for kinase therapies is about US$20 billion per annum, forecast to increase markedly. There are over 100 small-molecule kinase inhibitors active in late stages of clinical development and many more are likely to be approved in the coming years.
I see both positives and negatives to this – clearly, TKIs are an important target and their performance can likely be improved, but equally, with so much interest in the field, can a small biotech such as Theseus expect to develop class leading drug candidates in the face of such intense competition?
The second reason Theseus piques my interest is because the management team have successfully developed drugs that have gone on to be commercially successful in the past, and were able to build a company that ultimately became an acquisition target for a big pharma concern prepared to pay 75% above the traded share price to gain access to its drug candidates.
Can messrs Clackson, Shakespeare, Kerstein and Rivers repeat the trick with Theseus? The company certainly has some well-known venture capital backers, including OrbiMed – which seeded the company and then span it out into its own entity – Boxer Capital, and Foresite Capital – which led the company’s $100m Series B private funding round.
Theseus’ share price has been on a sharp upward trajectory so far in 2023, gaining >170% across the past month, although the only public communication from the company in 2023 so far has been a press release outlining “key anticipated 2023 milestones,” and announcing its third development program, BCR-ABL, which targets hematological cancers Chronic myeloid leukaemia (“CML”) and Acute Lymphoblastic Lymphoma (“ALL”).
Will we see further upside in 2023, now that the company’s market cap has risen >$500m, or could the gains be short-lived? In this post I’ll take a closer look at the rationale behind each drug program and try to answer that question.
THE-630 Targeting GIST – The Only Candidate In The Clinic
Usually, most of an early stage biotech’s value is tied up in its lead candidate and Theseus’ corporate presentation teases the fact that THE-630 targets gastrointestinal stromal tumor (“GIST”), a form of stomach cancer, and a potential blockbuster (>$1bn revenues per annum) opportunity for the company.
Specifically, THE-630 targets the KIT gene and protein, altered forms of which are often present in different forms of cancer. According to Theseus, ~90% of relapsed GIST cases are associated with secondary resistance mutations in KIT.

treatment options for GIST – deficiencies (Theseus Presentation)
As we can see above, there are four separate therapeutic options developed by big pharma to treat GIST, but Theseus argues that none are quite satisfactory, an opinion that’s borne out by generally poor Overall Response Rates (“ORR”) and Progression Free Survival (“PFS”) durations.
Theseus says that its predictive resistance assay has predicted that THE-630 “would have pan-variant inhibition and could substantially improve outcomes”, and as evidence, has shown that its PRA system was able to identify, for example, which exon mutations would be covered by Sunitinib and Ripretinib respectively, i.e. Sunitinib in exon 11 + 17/18 subgroup had an ORR of 0%, whilst Ripretinib had an ORR of 44% in the same subgroup in their respective clinical studies – much higher than its average ORR.
It’s Theseus’ contention that THE-630 could cover all mutations, unlike the approved therapies, as shown below.
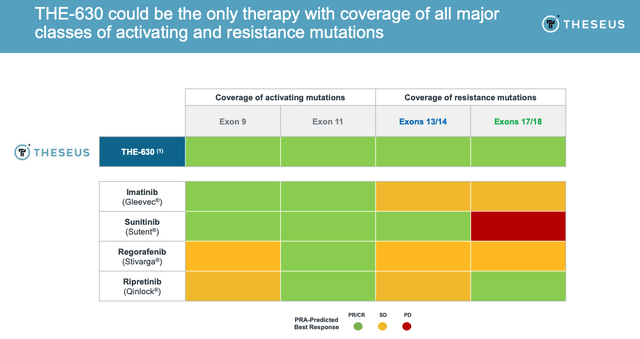
THE-630 predicted coverage versus approved therapies (Theseus Presentation)
Of course, Theseus now has to prove that thesis in the clinic, which will be no easy task. The Phase 1/2 study will enroll:
Patients with advanced GIST who have had disease progression on or are intolerant to imatinib therapy and have also had at least 1 of the following: sunitinib, regorafenib, ripretinib, or avapritinib
The initial Phase 1 endpoints are safety related – the drug will not progress beyond Phase 1 if its toxicology profile does not check out – but the Phase 2 dose expansion cohorts will attempt to establish the anti-tumor activity of THE-630 with a primary endpoint of confirmed ORR.
Theseus hopes it will be able to rapidly advance into registrational studies with THE-630 particularly evaluated as a 2nd or 5th line therapy. Ordinarily, investors would probably not look that far ahead, but with Theseus’ Senior Management team having successfully developed three TKIs in the past that are now approved – Exkivity being a third Takeda / Ariad candidate that is targeting peak sales of up to $600m per annum in NSCLC with EGFR exon 20 insertion mutations.
THE-349 – A Next-Generation EGFR inhibitor in NSCLC?
Speaking of NSCLC, that is the ultimate target of Theseus’ next most advanced candidate THE-349, which will look to target “18,000 first-line EGFRm NSCLC patients in the United States with mutations in exons 19 or 21,” according to Theseus.
The standard of care is currently AstraZeneca’s Tagrisso, a >$5bn per annum selling asset, but as with THE-630, Thesues believes THE-349 can handle a broader scope of mutational coverage than Tagrisso (osimertib), with preclinical studies showing the candidate “demonstrates tumor regressions against variants associated with both 1st and 2nd line osimertinib failure.”
THE-349 still requires its IND to be submitted and FDA approved, but Phase 1a and 1b dose escalation studies are already being planned, and expansion populations identified. The opportunity here, as with THE-630, is as a second line therapy, Theseus believes, and once again, management is eyeing a >$1bn market opportunity.
BCR-ABL Program – Driving The Share Price Upside?
Is it a coincidence that Theseus’ share price started climbing rapidly as soon as the latest program was announced?

BCR-ABL versus approved therapies in CML / ALL (Theseus Presentation)
Similarly to THE-630 and THE-349 Theseus believes BCR-ABL can succeed in a broader range of areas where currently approved therapies cannot – including Iclusig, which its management team helped develop.
It’s certainly true that treatment of hematological cancers has made great strides in recent years, with cell therapies in particular driving impressively high response rates, including complete responses. Theseus states, however, that:
in newly diagnosed patients treated in combination therapy with 1st or 2nd generation TKIs, relapse is associated with BCR-ABL resistance mutations in up to 75% of patients
The addition of TKI’s to cell therapy and chemotherapy can help improve outcomes, but it’s Theseus’ contention that its candidate can drive better efficacy with less toxicity, and potentially reduce the need for hematopoietic stem cell therapy (“HSCT”), which remains a risky therapy that’s tough on patients.
Conclusion – Theseus Recent Gains May Be Mysterious But There Is Genuine Potential In The Pipeline – I’d Consider Buying A Dip
With a share float of only 38m perhaps its is not a surprise that Theseus stock is capable of significant volatility, which may explain the stock’s recent gains – small buys will trigger a surge in the price.
With shares now trading only slightly below the IPO price, they’re not necessarily overvalued relative to the opportunity on offer – the chance to back a company whose management team has successfully developed three TKI therapeutics driving sales in the triple-digit millions, who played a key role in the >$5bn buyout of former company Ariad, and who are targeting blockbuster market opportunities.
The problems Theseus has identified with existing TKI in the fields of stomach, lung and blood cancers and its strategy for overcoming these issues seems persuasive on paper, and based on management’s experience, and there is preclinical data to back this up.
In 2023, Theseus will enter the clinic and before the end of the year we will have clinical data to make a much clearer judgment as to whether the theory can work in practice. With >$220m cash and a net loss of $37m in 2022, funding should not be a problem, and even if THE-630 disappoints in the clinic, the share price may not be devastated since there are two more programs targeting blockbuster market opportunities.
Without a clearer explanation as to why shares have picked up so suddenly in 2023 I would be unwilling to buy at current price, but since biotechs have a habit of losing value when there are no near-term catalysts in play, if Theseus stock drops <$10 per share in H1223, I’d be tempted to make a speculative buy based on the first clinical data from the THE-630 program.
The prospect of Theseus being an M&A target is also a tantalizing one for investors to contemplate. I plan to monitor Theseus closely this year and see if the company really can develop the next generation of TKIs, and reap the substantial rewards that would come with such progress.


