Immunome: Clinical Data With A COVID Therapeutic And The AbbVie Collaboration (IMNM)
ClaudioVentrella
Immunome (NASDAQ:IMNM), a biopharmaceutical company that develops antibody therapeutics using its proprietary discovery engine, entered the clinic in 2022 with its triple antibody cocktail therapeutic for COVID (IMM-BCP-01). The first clinical data from IMM-BCP-01, reported in January 2023, and a new collaboration agreement with AbbVie (ABBV), are the focus of this article.
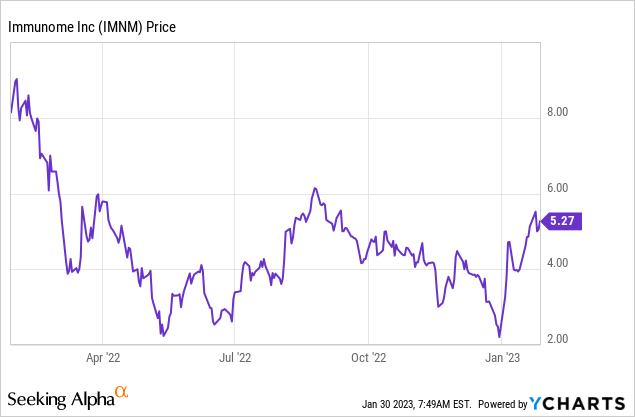
Figure 1: Past 12 months of trading activity for IMNM.
IMM-BCP-01: An antibody cocktail for COVID-19
An often mentioned pipeline candidate in 2022
Throughout 2022, IMNM issued several press releases regarding IMM-BCP-01, including some reporting that IMM-BCP-01 had neutralizing activity against many variants of SARS-CoV-2. As such, the phase 1b study of IMM-BCP-01 that got underway in June, 2022, and its eventual readout seemed like an important catalyst for IMNM. Indeed when IMNM reported financial results on November 14, 2022, IMM-BCP-01 was the first drug mentioned by the CEO.
We look forward to completing our clinical trial for IMM-BCP-01, our COVID-19 antibody program, while continuing to expand our oncology portfolio including our planned submission of the IND for IMM-ONC-01 by mid-2023. Our Discovery Engine provides unique insights into how the human immune system sees disease and continues to highlight interesting areas of cancer biology and identify novel targets and corresponding antibodies.
Dr Purnanand Sarma, President and CEO of IMNM.
Further, IMNM’s November corporate presentation notes that IMM-BCP-01 is being developed in collaboration with the US Department of Defense and the Defense Health Agency, and had been awarded up to $17.6M in funding (Contract number: W911QY-20-9-0019). IMM-BCP-01 is also the first drug presented in detail in the November 2022 corporate presentation, and indeed it was the only drug IMNM had in trials. For such a key part of the company’s pipeline and something the company was looking forward to completing a trial of, I would have hoped the company would put out a press release.
The phase 1b data so far
The IMM-BCP-01 phase 1b data is found on slide 33 of 34 of the January 2023 corporate presentation, and no press release was issued. A single dose of the cocktail (600 mg total, n = 6) produced data showing that IMM20184 and IMM20190, two of the antibodies found in IMM-BCP-01, have an apparent half-life of approximately 23 days. For the third antibody found in the cocktail, IMM20253, there was an apparent half life of 11 days, but that antibody “appears to exhibit enhanced antigen dependent PK [pharmacokinetics] at the dose administered (slide 33).” To be clear, the phase 1b study enrolled patients with mild to moderate COVID-19 and so the antigen (the spike protein, to which IMM20253 binds) is actually found in these patients.
Further, no significant treatment-related adverse events were reported which is good news, although it seems only so many patients were dosed with the cocktail so far. Three patients were treated with placebo, in keeping with the 2:1 randomization specified in the trial protocol. To be fair, the trial only has an estimated enrolment of 36 patients, and with a single ascending dose design, other patients may have been dosed with lower levels of the antibody cocktail than the six patients mentioned, which can nonetheless provide some additional safety data, or there may have only been nine patients in the trial so far.
Clinicaltrials.gov NCT05429021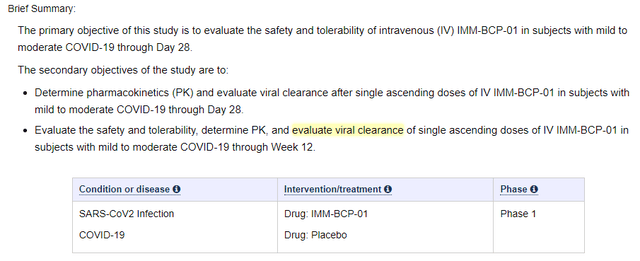
Lastly, while these initial results are mostly focused on safety/tolerability and IMNM has told us about the pharmacokinetics of the drug too, there were no data presented on the viral clearance properties of the drug. Evaluation of viral clearance was a secondary objective of the study according to the clinicaltrials.gov entry concerning the phase 1b study, but at the same time when IMNM is talking about six treatment vs three placebo patients, we can’t really expect too much in that regard. Data on viral clearance from six vs three patients would not be very reliable.
IMNM’s January corporate presentation also notes that “We completed dosing of the first cohort of patients with no significant treatment-related adverse events. We have decided to seek a partner in order to continue the trial and for any further development activities (slide 10).” IMNM then might dose another cohort of patients with a partner at which point we might get something more concrete in the way of initial hints of efficacy (viral clearance for example).
The ABBV deal
On January 6, 2023, IMNM noted it had struck a deal with ABBV to identify up to 10 novel target-antibody pairs arising from three specified tumor types using IMNM’s discovery engine. That ABBV is interested in IMNM’s antibody discovery engine is a nice vote of confidence in IMNM’s technology.
… Immunome’s approach has the potential to unlock novel cancer biology and yield multiple therapeutic candidates. We look forward to utilizing their Discovery Engine to enhance our existing oncology pipeline.
Steve Davidsen, Vice President, Oncology Discovery Research, ABBV, January 6, press release.
The upfront payment of $30M is a nice detail of the deal and IMNM can also receive platform access payments up to $70M if ABBV elects to continue research with IMNM’s discovery engine. Further, IMNM is eligible to receive development and first sale milestones of up to $120M per target, with potential for further sales-based milestones and royalties on sales.
Conclusion and Financial Summary
As of September 30, 2022, IMNM reported cash and cash equivalents of $27.1M, although $30M upfront from ABBV would bring pro forma cash to $57.1M. Net loss for Q3’22 was $8.5M and net cash used in operating activities was $22M in the first nine months of 2022. At that rate IMNM would burn through $57.1M in 23 months, which starting at Q4’22 would get them into Q3’24. Consistent with my calculation, IMNM notes in its January corporate presentation that it has cash at least through 3Q’24. This is of course assuming no further payments from ABBV in that time, which would be a non-dilutive source of more cash. There were 12,128,843 shares of IMNM’s common stock outstanding as of November 10, 2022, corresponding to a market cap of $63.92M ($5.27 per share).
There is not much not to like about the ABBV deal, I can see why IMNM discusses this development first in the recent corporate presentation and why IMM-BCP-01 didn’t even get a press release. Further, IMM-BCP-01 might appeal to a potential partner, but the ABBV deal is obviously worth highlighting for the company. I consider IMNM a strong buy given it is only trading at cash, the ABBV deal is a major positive development, not only in terms of the cash it can bring in, but the vote of confidence it provides in IMNM’s discovery engine. IMNM still has additional catalysts outside of potential payments from ABBV as IMNM discovers target-antibody pairs for ABBV. For example IMM-ONC-01, an antibody targeting IL-38 with potential in multiple solid tumors could enter the clinic in 2023 as IMNM notes an Investigational New Drug [IND] submission is expected in 2023.
The risks of any long in IMNM are several fold, a few of which are mentioned here. Firstly, delays in producing target-antibody pairs, which ABBV exercises its options to develop, could be seen as negative and cause the share price to fall. Further, delays in the IND submission of IMM-ONC-01 could cause the share price to fall, as could any clinical hold. IMNM previously had a clinical hold of IMM-BCP-01 following IND submission which led to a delay. I’m not expecting this with IMM-ONC-01, but it is always a possibility with any drug. Lastly, time passing with no update on IMM-BCP-01, such as no partner being found, could lead to some writing off the value of that drug.
Editor’s Note: This article covers one or more microcap stocks. Please be aware of the risks associated with these stocks.


